If CBD manufacturers are sending their materials to credible 3rd party CBD testing facilities up to three times during the process, how is it that sometimes when a third-party lab tests, they get a different result?
Analytical Chemistry Lesson
Before we can get too far, we need to understand a little bit about how lab testing works. In order to tell how much of something is present, we need a way to separate it out from the others. We then need a way to tell how much is there.
Back in the early 1900s, a Russian botanist, Mikhail Tswett, was trying to figure out what was in various tree leaves that gave them color. He came up with a totally new idea. He ground up the leaves, and extracted them in a solvent. Next, he filled two columns with different materials (chalk = calcium carbonate, and alumina). He then poured the extract over the columns, and followed that by more solvent. What he saw was that some of the material began to separate out as they passed through the column. He coined the phrase “chromatography” – color separation. It might have looked something like this:
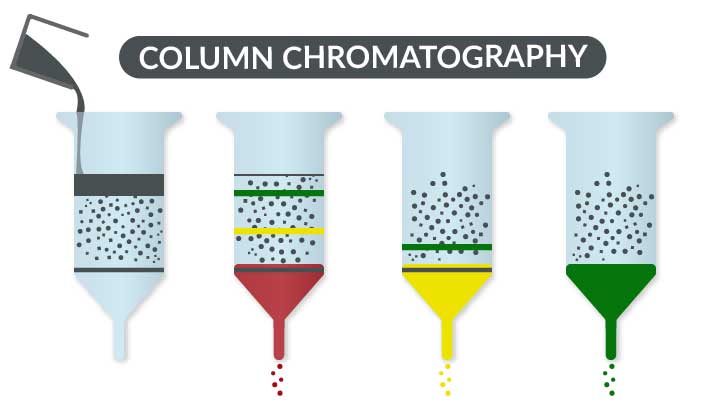
Over the years, scientists have discovered that chemicals can typically be separated in one of three basic ways: size/weight, charge, and “oiliness”. The columns are the main thing that helps separate the materials. Some have charges which allow separation based on the charge of the chemical. Others have tiny pebbles that help separate based on size. While others have long hydrocarbon chains which allow the “oily” particles to react. This “oiliness” is officially known as hydrophobicity (aka afraid of water) or its flip hydrophilicity (aka friend of water). In each case, whether size, charge or “oiliness”, the interaction between the item and the column causes some things in the mix to go through faster than others.
If you put a detector at the bottom of the column, you then can trace the materials as they come through. How long it takes to come through is a way to identify what chemical it is. How tall the peak is, or how much area is under the curve helps the analyst figure out how much of that chemical is there.
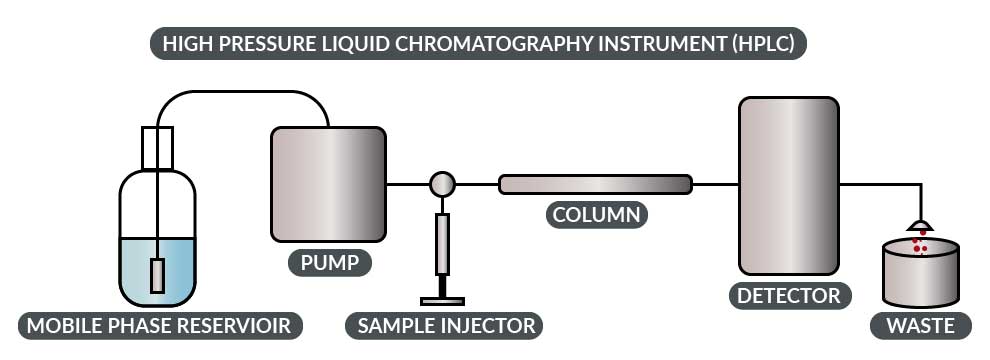
The primary workhorse for this testing is the High Pressure Liquid Chromatography instrument (or HPLC). An HPLC has these basic parts: injector, pump, column and detector. In order to make an analytical run, you set up the column and pumps by pumping a liquid through it. Exactly which liquid is used depends on which separation you are trying to achieve and what the column requirements are.
Once the system is set up, you inject your sample and observe the “chromatogram”, which is the trace of the different chemicals as they are separated and pass through the column and go past the detector. You would probably see something like this:
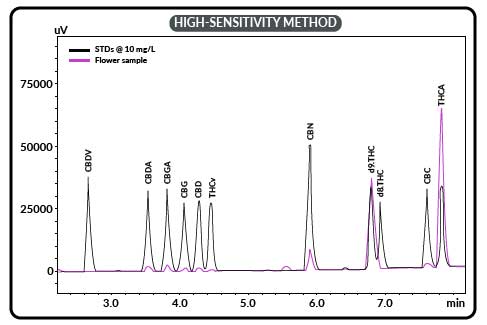
In this case, they were running a CBD oil standard (black) against a flower extract (pink). You’ll notice that the time runs across the bottom, and the absorbance run up the side. This is a way that you can see how much there is from one peak to another, as well as which peak is what (based on the time to get to the detector).
These are actually two different runs that are over-laid on top of each other to make comparison easy. Without really knowing anything, you can see that there is much more pink THCA in the flower than the standard. With the exception of d9-THC where the flower and standard have about the same, all the other items have much more in the standard.
From above you know that we need to know the area under the curve to figure out how much is there. Fortunately, the machines of today can easily tell us this once we tell them where to start and stop (based on times, or manually entering that information).
At this point, we need to have a standard for comparison. If we have a pure standard (say pure CBD), we can then say that in this instrument, under these conditions, 1 mg of pure standard gives a peak area of XXXXX.
You know the two areas (they are given by the machine printout) and you know the weight and purity of your standard (yes, you do need to adjust for the purity – more on that later). So, you just do simple math to solve for the weight of the unknown. Now you know how many mg of that chemical (we’re using CBD as the example here) are in the sample. You can back-calculate using the volume injected to get to mg CBD/ml of oil, or weight of plant extract or however the sample was delivered.
Above is a simple example of how it works. Now, let’s delve into a little more detail as to why there might be some issues between suppliers and labs.
Method
How well the different parts are separated depends on the method. This includes the column, the solvents used, and the detectors used. Each of these can play a part in that area under the curve number.
It is theoretically possible to separate CBD oil components using any one of the three possible strategies mentioned (charge, size, oiliness). However, some are better than others at separating all the parts, particularly of something as complex as full spectrum CBD oil.
Once the strategy is decided, then the column must be selected. There are a wide variety of columns capable of performing each of these three strategies. The specific column selected will go a long way to determine the time it takes from injector to detector as well as what that area curve looks like. Our example is a very nice and tight peak. Depending on the column selected, it could broaden out and make it more difficult to determine starting and stopping points for that specific peak.
After the column is selected, then the method is fine-tuned by determining the specific solvents that will be used. Just like the column selection, the solvent selection can impact retention time and the overall peak shape and height. Similarly, the pressure that is run (and the pump speed to achieve that) are also impactful of that final peak.
As a point of fact, most HPLC methods are considered “good” if they can run the same sample 5-10 times in a row and the variation between the samples is less than 1%.
Part of the issue for CBD related testing is that there are no internationally approved standard methods. Using pharmaceuticals and officially recognized nutritional supplements as the example, there are various international bodies that “certify” a method. In the United States, one typically finds that United States Pharmacopeia (USP) will list a specific method that should be used for testing a substance. If you choose not to use that method, then it is expected that you will show that your method is equivalent or better than the method published in the USP.
If the labs do not have identical methods (and sometimes even identical machines), then there is risk that the results will not exactly match.
Standard
Now let’s talk about the standard itself. If you are trying to test a CBD oil, then you need multiple standards. For example in the chromatogram above, there are peaks for CBD, CBG, CBN and THC, as well as their variations (CBDA for acidic CBD and so on). If you want to quantitate, you need a standard for each one of these.
Earlier, we mentioned the importance of purity. In order to calculate back from the area under the curve to the amount in your sample, we use the simple ratio described. However, purity impacts the weight of the sample. For example if you weigh out an amount, say 1 pound, of 100% pure then you will have exactly 1 pound of it. If you weigh out 1 pound of something that is 98% pure, then you have 0.98 pounds of that, and 0.2 pounds of something else. The best analytical methods need to include purity of the standards in order to say exactly how much is in the unknown sample.
Part of the issue for variation between one test and another (this is true for any analytical test, not just CBD related) is that there must be agreement as to what 100% pure is in order to compare one lab to another. For example, if my lab makes a “100% pure” standard and all of our data is calculated using that ratio, we are comparing everything to that standard. If another lab makes their own standard, then likewise all of their data is compared to that standard. Now, what if my standard when run against your standard is not 100%? Then our results cannot be the same as we are not comparing to the same thing.
Now, consider how that works in the case of CBD oil. There could be as many as 11 standards needed for that chromatogram above (1 for each peak) if you want to calculate the amount of them all in your sample. If each of those standards vary by some percent one lab to another, then it becomes very difficult to compare the entire data set.
In an attempt to address this, you may see instead of an exact mg amount in the test sample that a percent is reported. To do this, they add all the areas under the curve, and call that 100%. Then each individual curve can be set up as a percent ratio to determine its contribution to the total. In this way you can estimate the percent of total that the CBD curve represents. Often labs using this method do it on a single injection, which is not nearly as accurate as averaging across several injections (which is what is done when you are using a standard to determine an amount).
Part of the issue for CBD related testing is that there are no internationally recognized standards. Again using pharmaceuticals and officially recognized nutritional supplements as the example, there are various international bodies that “certify” a standard. In the United States, one typically finds that USP sells certified standards which tie to internationally recognized standards. This is done to alleviate the possibility of variation of results due to variation between standards used when testing.
To achieve consistent results between labs, the standards used must be as close to identical as possible, and international certification is the best way to achieve that.
Sample Prep Issues
Now let’s look at the challenge of the sample itself. Remember that a lab is testing the plant itself, the in-process samples during purification/production, the material in ready to fill bulk, and sometimes from filled product. Each of those represents some different challenges.
We already talked about method variation, and how that can impact results. That is true when you have exactly the same sample being injected. But, what if you have to inject different kinds of samples? What you need to do to plant leaf material to get CBD related chemicals into a testable solution, is different that of the purified material, and different still from something out of a bottle.
You likely need to grind up plant materials, and extract with solvents in a very similar way to our Russian friend above. Each solvent you choose can impact how it flows onto and over the column. In addition, the concentration can impact it as well. So, the method needs to be adjusted for these kinds of samples in order to get the most accurate results.
Pure CBD can be a gooey ball. Trying to get a good representative sample from this and then get it into solution is challenge. Things on the outside of the ball may have been oxidized or dried out compared to things on the inside. In addition, there are some of the same solvent challenges as you find in plants – how to get it all in solution and injectable. Again, these can cause a need to alter the method.
Finally, the bottled material can come in different forms. Vape oils might have glycols in them to aid suspension. Sublingual products have different carrier oils (hemp seed oil, MCT, olive oil are often used). Each of those sample details present different sample prep issues that can cause the method to need to be adapted.
The method used for pure standards often has to be adapted for the reality of the specific sample and this (sample prep method) can lead to variation between lab results.
What does all of this mean?
Historically, the FDA has taken a dim view of products that claim to do many wonderful things with little or no scientific proof. Unfortunately, that now includes CBD, at least that is what Scott Gottlieb, the commissioner of the FDA has been saying, and directly said the same day the President signed the 2018 Farm Bill into law. What that means is that these products are directly in the cross-hairs of the FDA – despite the passage of the 2018 Farm Bill. In the past several years, the FDA has made statements regarding the “fact” that few final products met within 10% their label claims for CBD content, and has written warning letters to various manufacturers stating that their products do not meet label claims.
Of course, everyone assumes that the FDA knows how to test (and of course they do), but without an agreed upon standard method with defined sample preps and an internationally recognized standard, their numbers are as suspect as anyone else!
Let’s be clear here. The issues described above are enough to explain having trouble agreeing at the plus and minus 10% of label claim level. It might even cover plus and minus 15 or even 20%, but that’s stretching it to say the least. Some of the manufacturers were at less than 10% of label claim which is far different than within 10% of label claim. Testing differences will not explain that kind of difference – those were shady manufacturers!
In an attempt to shore up various issues, the US Hemp Round Table is putting forth a new independent certification. This is called the US Hemp authority. The intent is to certify manufacturers to have GMP practices and quality systems. Once certified, those manufacturers can display a logo which suggests that the manufacturer has a very high quality from start to finish. They do have some defined methods based on AOAC (Association of Official Agricultural Chemists) methods and commercially available research chemicals. This is a step in the right direction, but more scientific rigor and detail is needed.
The best path for the industry to take is to develop an agreed upon standard method, and a reference standard that all agree to use as a reference (or that various standards we see above). Several agencies have been said to be working toward this including ASTM International (formerly known as ASTM or the American Society for Testing and Materials), NIST (National Institute of Standards and Technology) and likely even USP itself.
The USP has built practices which allow for new products to be introduced. There are supplement chapters, and there are biologics chapters as well. For any given chemical with a chapter in the USP there are a variety of tests defined that must be performed in order to sell the product and utilize the USP logo on the label. Because of the FDA stance regarding the API (aka active pharmaceutical ingredient, such as THC and CBD in recently approved drugs) not allowing CBD to be offered as a supplement, it may be politically difficult to get chapters related to CBD approved through the USP process. It would be an excellent place for organizations such as the US Hemp Authority and US Hemp Roundtable to lobby the USP to accept chapters on CBD oil and similar products so that any questions of legality and testing variation could finally and ultimately be addressed.
Bottom Line
Variations in sample prep, analytical methods and standards used for testing can cause significant differences in results from test to test and more so lab to lab. The industry needs to agree to what the “official” method and standards are before we can truly independently and accurately compare what’s on the label to what’s in the bottle.
Rodney Zollars, Skollarz LLC
Latest posts by Rodney Zollars, Skollarz LLC (see all)
- CBD Testing – What’s the Big Deal? - February 27, 2019











